
Would you eat a McDonalds Big Mac that was out-of-date, had been dropped on the floor, and made with a stranger’s bare hands?
No! Me neither.
Unfortunately, that’s what thousands of people in China, unknowingly, did.
Back in 2015, the Chinese meat suppliers for McDonald’s were secretly filmed and caught:
“Handling meat and chicken with their bare hands, taking meat that had fallen on the floor and adding it back on to the production line…and forging production dates on beef patties.” – ET2C, Lessons on the Importance of Quality Control Checks
Around 4,300 McDonalds beef patties were tested and found to be out-of-date and contaminated with bacteria.
As a result, over 2,000 McDonald’s outlets across Asia were dramatically affected. The reputation of McDonald’s and their meat supplier was irreversibly damaged, and McDonald’s saw a 7.3% drop in sales.
McDonald’s was definitely not ‘lovin it’.
If only they’d used Enterprise Quality Management Software (EQMS) to regulate their supplier’s meat handling processes and control the quality of the produce they were delivering.
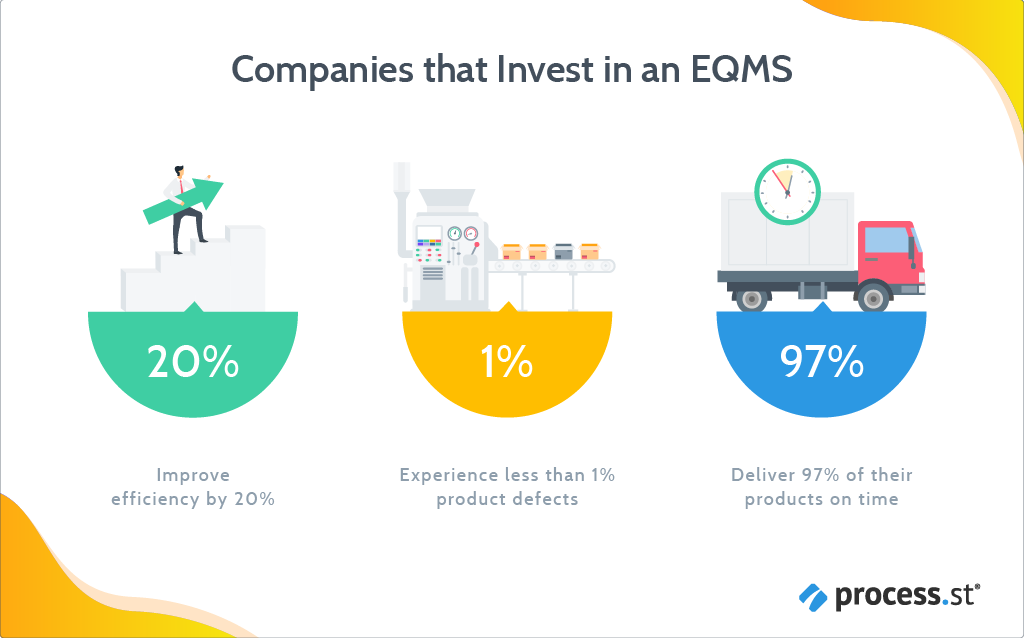
Companies who use EQMS to manage quality, experience fewer product quality issues (less than 1%), and improve their productivity by 20%.
Keen to find out more? Join me in this Process Street post as we run through the following:
- What is EQMS and how does it work?
- Who uses EQMS?
- Is EQMS it just for regulated organizations?
- What are the bottlenecks of EQMS?
- The best EQMS alternative (+free templates)?
- Top 8 EQMS software
Let’s make our way through the golden arches and get started!
What is EQMS and how does it work?

In its simplest form, EQMS (Enterprise Quality Management Software) is a technical solution to help organizations manage the quality of what they’re producing.
It’s used by enterprises to make sure they follow industry regulations, observe best practices, and consistently produce high-quality products that keep their customers happy.
How does EQMS do that?
EQMS, as the name suggests, is a piece of quality management software that joins an organization’s quality control processes together, and collects and displays key quality control data from those processes. Data on metrics such as the number of inaccuracies, maintenance frequencies, and speed of production enables organizations to spot issues, maintain regulatory compliance, and keep quality levels consistently high.
“EQMS centralizes, standardizes, and streamlines quality content and processes throughout the value chain, enabling cross-functional communication and collaboration on critical issues” – CDN2, Unlocking the Value of Enterprise Quality Management Software
Who uses EQMS?
Any organization that gives a damn about quality would benefit from implementing EQMS. However, only 20% of companies are currently using an EQMS to control and manage quality. It’s generally only adopted by corporations that are heavily regulated. Companies in the food, financial or aerospace industries, for example, that need to be compliant with industry standards and regulations such as ISO 22000, AS9100, or ISO 9001.
“To stay competitive, more and more organizations are turning to EQMS to meet quality standards and maintain compliance with quality regulations imposed by the FDA, ISO, EMEA, and other similar regulatory bodies.” – MasterControl, Enterprise Quality Management Software Systems (EQMS)
Why do regulated companies turn to EQMS?

Regulated companies, like JP Morgan or Boeing for instance, face the same battles that non-regulated companies face. They’re under constant pressure to make a profit, stay competitive, and cut costs. But on top of all that, regulated companies also face strict regulations, frequent audits, and hefty fines if they fail to meet regulatory expectations.
Maintaining compliance while trying to run a successful business is difficult for companies that rely on outmoded tools, manual processes, and a fragmented approach to quality management.
Regulated companies need a reliable system that saves them time and takes the stress out of managing quality. They need something that can automate, streamline, and connect all their quality processes, so they can focus on accelerating speed to market, reducing costs, improving efficiencies, minimizing risk, meeting customer requirements, and ensuring compliance.
But what if you’re not in a heavily regulated industry? Is EQMS the right solution for you?
Is EQMS just for regulated organizations?
I’ve got a couple of questions for you.
1. What makes a business successful?
Is it:
- a) Keeping customers happy
- b) Crushing the competition
- c) Efficient, error-free manufacturing processes and cost reductions
Of course, it’s all three, right?
2. And how do you make sure you achieve all three and, therefore, become a successful business?
Is it:
-
- a) Focus on quality
-
- b) Focus on quality
- c) Focus on quality
I think you’ve got it! To keep your customers happy, your competition at bay, and your manufacturing processes efficient, error-free, and cost-effective – you need to focus on quality.
Which is where EQMS comes in.
Not only is EQMS a single source that makes it easier for organizations to stick to standards and regulatory requirements, it also allows them to:
- Monitor and control the entire approach to quality management, from one single platform
- Assess how their organization is performing as a whole, in real-time
- Identify and resolve bottlenecks and pain points in their design and production methods
- Make informed decisions around quality management processes
- Standardize operations across the entire organization
- Achieve operational excellence throughout the product life cycle
Investing in EQMS allows organizations to control and manage quality quickly, easily, and with less risk. It allows them to make sure their customers are happy, their processes are efficient, and competition is outpaced.
“Not having an EQMS creates risk by forcing your company to rely on manual processes. Ultimately, not implementing EQMS costs manufacturers money by encouraging a reactive approach to quality.” – CEBOS, The Risks and Costs of Not Having EQMS
If your company is dealing with time-consuming paper processes, disparate databases, or poor supplier quality, you should consider implementing Enterprise Quality Management Software.
But, wait a second.
“While more companies are talking about adopting an EQMS system, few are making progress when it comes to actually budgeting for and implementing such a system.” – PharmaManufacturing, Don’t Slow Your EQMS Adoption Roll
What are the bottlenecks of EQMS?

As great as EQMS sounds, there are a few barriers to implementation that we need to consider.
EQMS downfall # 1: It’s overly expensive & overly complicated
There are so many EQMS solutions out there, all offering different benefits at varying prices. But, to give you a general idea, implementing a standard, robust EQMS system, can cost you anywhere from $30,000 to $75,000 per year.
But it’s not only the annual cost you have to consider.
EQMS systems are also renowned for being complex, burdensome, and difficult to use. In fact, a recent Deloitte study identified that some manufacturers are spending up to $100,000 and 116 workdays per year trying to comply with overly complex Enterprise Quality Management Software systems.
So, EQMS can be costly, both in terms of initial implementation and upkeep, and also everyday usage.
EQMS downfall # 2: It takes a long time to implement
The implementation of new software into an organization is often filled with teething problems and unforeseen complications. It can be a steep learning curve for all involved. This is especially true for larger enterprise organizations, and implementing an EQMS solution is no exception. Training management first is essential.
The installation of EQMS systems often exceeds the expected delivery time and organizations can encounter numerous issues and frustrations as they adapt to the new way of working, which can sometimes lead to customer complaints as product development slows as a result.
EQMS downfall # 3: One size does not fit all
Many organizations will willingly spend the time and money implementing an all-singing, all-dancing EQMS system because they’re under the impression that it will revolutionize everything.
But, as many EQMS systems come as out-the-box solutions (unless you invest a lot of time and money in a custom platform), organizations are often left with an incredible piece of software that is completely above and beyond what they need.
If the EQMS solution doesn’t fit with organizational goals, budget, structure, or stage of growth, companies are likely to waste money, confuse their workforce, and be left with a complex tool that loses any and all actionability as a result.
“A system is only as good as how its users can proficiently utilize it for various operations.” – CloudSecureTech, What is EQMS (Enterprise Quality Management Software)?
To summarize, although the benefits of having an EQMS system make it a must-have for many large regulated enterprise organizations, EQMS does have some hefty downfalls. It can be complicated and expensive to use, it requires lots of training, and it can take a long time to implement and become beneficial to organizations.
The best EQMS alternative (+free templates)
There is a great alternative to dedicates EQMS software
“A narrow focus on ‘hard’ quality management tools can be at the expense of the support of ‘soft’ quality management practices, which can help to more effectively embed a ‘culture of quality’ across an organization” – Cognidox, QMS, eQMS or BMS? What’s the Difference?
If the hardcore, complex EQMS systems don’t feel like the right solution for you, there is another option.
Process Street is a powerful yet easy to learn no-code tool that allows you to create, document, monitor, and manage all the processes within your organization. Basically, it’s super-powered checklists.
If a quality management system allows you to oversee your inputs and outputs in terms of a quantifiable “quality”, Process Street allows you to track and manage all of your internal processes in the form of checklist templates. You can effortlessly build and maintain all of your internal procedures, and see who is working on them in real-time. Rich form fields mean you can track detailed data and information and have the results of each process output to a spreadsheet or .PDF via one of thousands of integrations.
It will still help you maintain industry standards, risk management, maintain a standard of quality, and meet customer expectations while also increase operational efficiency.
Before I go any further, watch this video from managed services provider TechMD to hear about how they use Process Street to manage their internal processes. You could also read this article to find out a little more about what Process Street is and how it can make your life easier.
Although it’s not an “official” EQMS system (whatever that is), Process Street can help you to achieve the same objectives as EQMS can, in terms of meeting standards of quality.
Take a look at the below ISO 9001 internal audit process for quality management systems as an example. This process template was created to help organizations meet ISO 9001:2013 audit criteria:
Click here to access the ISO 9001 Internal Audit Checklist for Quality Management Systems!
(Log in and add this template to your account, or sign up for a free trial, if you don’t have one yet).
So, as you can see, you can create templates for your quality control processes to help you standardize operations across the entire organization.
You can create tasks and assign them out to your workforce, to make sure they are following best practices, or meeting required standards as they work through their processes.
You can connect the process templates you create to the platforms you use regularly, via Zapier, Webhooks, or API integration. This means you can record, send, and store key data from your processes instantly and accurately.
The checklist dashboard will allow you to monitor all your quality management processes, from one central place. This will enable you to assess how your organization is performing as a whole, in real-time. It will also help you to identify and resolve bottlenecks in your design and production methods, and can inform your decisions around quality management processes.
And, to make life easier, you also have access to the Process Street template library, which is filled with thousands of pre-made quality control templates that you can use, for free.
Templates like these for example:
AS9100D Audit Checklist
Use this checklist as a framework to help you conduct an effective audit on the implementation of the AS9100D aerospace quality management standard.
Click here to access the AS9100D Audit Checklist!
FSSC 22000: ISO/TS 22002-1 Internal Audit Checklist
Use this checklist to help you meet the regulatory requirements set by pre-requisite programs such as ISO and 22002-1, and make sure you’re supplying safe food and beverages.
Click here to access the FSSC 22000: ISO/TS 22002-1 Internal Audit Checklist!
You can use any of the above templates as they are, or you can customize them. Use the below super-powered features to make your processes fit your specific organizational goals, objectives, structure, and industry:
- Stop tasks
- Dynamic due dates
- Task permissions
- Conditional logic
- Approval tasks ✔️
- Embed widget ️
- Role assignments ♀
- Webhooks ↩️
- Many, many more 🙂
Top 8 EQMS tools
Intellect
Intellect is a cutting-edge solution designed to enhance cognitive abilities and problem-solving skills. Leveraging advanced algorithms, this electronic quality management system facilitates efficient data processing, analysis, and decision-making. This powerful tool empowers users to optimize productivity and achieve smarter outcomes across various domains.
Key features:
- Document control: Centralized management of documents to ensure version control and accessibility.
- Audit management: Facilitates the planning, execution, and tracking of internal and external audits.
- Corrective and preventive actions (CAPA): Manages the identification, resolution, and prevention of non-conformities.
- Training management: Tracks employee training records and ensures compliance with certification requirements.
- Risk management: Identifies and assesses potential risks to quality and compliance.
Pros:
- It serves many industries, including cosmetics and medical device companies.
- Centralizes management of documents to ensure version control and accessibility.
- Helps organizations adhere to industry requirements and regulatory standards.
Cons:
- Initial costs for software purchase, training, and implementation can be high.
MasterControl
MasterControl is a leading provider of enterprise quality management software. Renowned for its robust solutions, MasterControl empowers organizations to streamline quality processes, enhance compliance, and foster innovation, ensuring efficiency and reliability in regulated industries.
Key features:
- Document control: Centralized management of documents with version control and access tracking.
- Quality event management: Facilitates the identification, investigation, and resolution of quality events.
- Audit management: Plans, executes, and tracks internal and external audits efficiently.
- Change control: Manages and documents changes to processes, products, or systems.
Pros:
- Captures and manages instances of non-compliance or deviations.
- Evaluates and monitors the quality of materials and services from suppliers.
- Ensures the security and authenticity of electronic records and approvals.
Cons:
- Some features take time to learn.
TrackWise
TrackWise, a quality management software by Sparta Systems, empowers organizations to streamline quality processes. Renowned for its robust capabilities, it ensures compliance, facilitates risk management, and enhances operational efficiency, contributing to the delivery of high-quality products and services in regulated industries.
Key features:
- Workflow automation: Automates repetitive processes for efficiency and consistency.
- Electronic batch records (EBR): Digitizes and automates documentation related to batch processing.
- Incident management: Captures and manages incidents and deviations from standard processes.
- Equipment calibration: Tracks and schedules equipment calibration to maintain accuracy.
Pros:
- Systematically addresses and prevents issues to improve overall quality.
- Manages and documents changes to processes, products, or systems.
- Identifies, assesses, and mitigates risks related to quality and compliance.
Cons:
- It doesn’t have as many features as other similar products.
Qualio
Qualio is a quality management software designed for life sciences companies. Offering compliance with industry regulations, Qualio streamlines document control, training management, and other quality processes to ensure efficient operations and adherence to regulatory standards in the pharmaceutical and medical device industries.
Key features:
- Data import/export: Facilitates the import and export of data for integration with other systems.
- Regulatory reporting templates: Offers predefined templates for regulatory reporting.
- Decision-making tools: Facilitates collaboration in decision-making processes.
- Regulatory document submission: Streamlines the preparation and submission of regulatory documents.
Pros:
- Supports multiple languages to accommodate global users.
- Ensures the QMS tool functions across different web browsers.
- Plans and executes validation activities to ensure software reliability.
Cons:
- It doesn’t have an equipment module.
Ideagen Quality Management
Ideagen’s Quality Management software is a robust solution designed to enhance quality processes. Offering features such as document control, audit management, and compliance tracking, Ideagen supports organizations in maintaining high standards, achieving regulatory compliance, and fostering continuous improvement in quality management practices.
Key features:
- User training modules: Provides modules for training users on the QMS tool.
- Compliance dashboard: Presents compliance status and metrics in a visual and easily digestible format.
- Trend analysis: Analyzes trends in quality metrics to identify areas for improvement.
- User role management: Defines roles and permissions to ensure data security and access control.
Pros:
- Helps to reduce the time to market.
- Makes life much easier for quality leaders.
- Enables tracking of products and materials throughout the supply chain.
Cons:
- They don’t do the best job updating their training materials.
Greenlight Guru
Greenlight Guru is a leading electronic quality management software software designed specifically for the medical device industry. It streamlines regulatory compliance, document control, risk management, and product development, enabling organizations to bring safe and innovative medical devices to market efficiently.
Key features:
- Validation planning: Plans and executes validation activities to ensure software reliability.
- Electronic signature: Ensures the security and authenticity of electronic records and approvals.
- Product lifecycle management: Manages the entire product development lifecycle with documentation and traceability.
- Regulatory compliance management: Ensures adherence to industry regulations and standards.
Pros:
- Evaluates and monitors the quality of materials and services from suppliers.
- Systematically addresses and prevents issues to improve overall quality.
- Centralized control and management of documents related to quality and compliance.
Cons:
- Users have to pay for the training content.
Arena
Arena is a comprehensive software solution designed to streamline quality processes in product development. It facilitates collaboration, document control, and compliance management, ensuring organizations in regulated industries deliver effective products efficiently and in adherence to industry standards.
Key features:
- Historical trend analysis: Analyzes historical data trends to predict future quality outcomes.
- Software validation tools: Provides tools to validate the QMS software for regulatory compliance.
- Data security measures: Implements robust security protocols to protect sensitive information and essential elements.
- Regulatory intelligence: Keeps organizations informed about changes in regulations and standards.
Pros:
- Maintains and organizes the documentation related to the design history of a product.
- Provides real-time visibility into quality and compliance metrics.
- Enables access to quality management functions via mobile devices.
Cons:
- The subscription and licensing fees are higher than most other similar tools.
ZenQMS
ZenQMS is a cloud-based quality management system designed for regulated industries. It streamlines document control, training management, and compliance processes, ensuring organizations maintain high-quality standards, adhere to regulations, and drive operational efficiency.
Key features:
- Nonconformance management: Captures and manages instances of non-compliance or deviations.
- Supplier quality management: Evaluates and monitors the quality of materials and services from suppliers.
- Validation and verification: Ensures the effectiveness and compliance of processes and controls.
- Customer complaints management: Captures, tracks, and addresses customer complaints systematically.
Pros:
- Connects with other business systems for seamless data exchange.
- Sends automated notifications and alerts for critical quality events.
- Provides predefined templates to facilitate compliance.
Cons:
- The audit trail features isn’t very robust.
Essential EQMS reading
You can find more posts like this, and the ones below, in our Process Street blog. Go ahead, fill your boots!
- ISO 9001: The Ultimate QMS Guide (Basics, Implementation, ISO Templates)
- Best QMS Software for Quality Management Systems: Which is Right for You?
- What is Quality Management? The Definitive QMS Guide (Free ISO 9001 Template)
- How to Use The Deming Cycle for Continuous Quality Improvement
- Quality Control: How to Get It Right With 19 Free Templates
- Total Quality Management (TQM): Improve Processes & Keep Customers Happy
Before we close out, I have one final observation I’d like to leave you with.
Remember earlier, when Deloitte established that some organizations were spending up to $100,000 and 116 workdays per year, trying to comply with overly complex, Enterprise Quality Management Systems?
Well, in another Deloitte survey, they found that if organizations simplified and centralized their quality management systems, they could reduce that to only 67 workdays and $51,000 per year.
Need I say more?

(P.s. McDonald’s are still using the same meat suppliers that they were in 2015, so I’d stay away from Big Mac’s in China if I were you!)
We’d love to hear about your experience with, or opinions about, EQMS. Talk to us in the comments below! Who knows? Your thoughts could get featured in an upcoming article!





